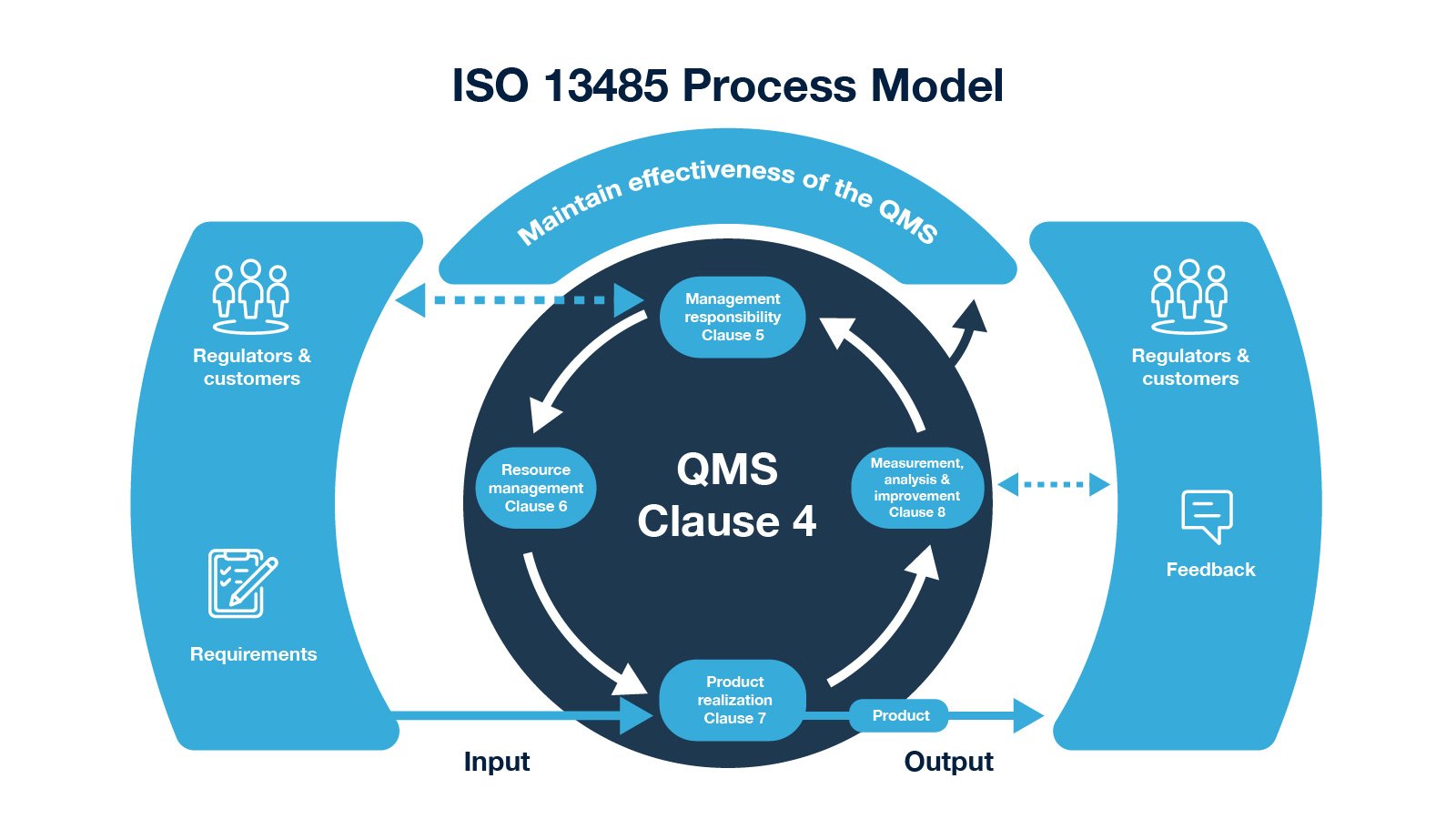
wishes to market its products internationally, it must comply with both the QSR and ISO 13485 manufacturing standards. Additionally, if a device maker based in the U.S. For the time being, separate guidance remains in effect.(2) Until the QSR’s shift to ISO 13485 requirements is fully completed, compliance with the QSR is required for manufacturers planning to distribute medical devices in the U.S. quality system requirements with ISO 13485, and plans to issue a notice of proposed rulemaking in October 2020. The FDA is in the process of harmonizing U.S. The QSR, also commonly called Current Good Manufacturing Practice ( CGMP) regulations, was established and is maintained by the U.S. How Does ISO 13485 Relate to the FDA’s QSR?įor device manufacturers eyeing American markets, the requirements of ISO 13485 standard can often seem blurred with those set forth in the 21 CFR Part 820 – Quality System Regulation (QSR). If conformance to both standards is necessary, the company must plan strategies for meeting each set of requirements. Additionally, while the two standards were once more harmonized, variations in their formats have occurred since ISO 9001 was restructured in 2015. It outlines additional requirements for documented procedures.ĭevice manufacturers can obtain certifications to both standards but may opt not to do so based on the intent of the two standards.It places more emphasis on risk management.ISO 13485 differs from ISO 9001 in two other significant ways: These include metrics related to meeting customer requirements and maintaining the effectiveness of the QMS. Rather than requiring medical device companies to meet the potentially subjective aspects of the ISO 9001 requirements, ISO 13485 is targeted toward meeting metrics that more accurately gauge quality performance. Although these are critical concerns for all manufacturers, they pose unique challenges for medical device makers because they tend to be too subjective and are therefore difficult to measure. Although both are in the same QMS family of standards, ISO 9001 is a general set of requirements that necessitates greater focus on continual improvement and customer satisfaction. ISO 13485 is a stand-alone document, but it was based on and is directly related to ISO 9001, the world’s leading quality management standard. The standard applies to all 165 member countries of the International Organization for Standardization (ISO).(1) How is ISO 13485 Different From ISO 9001? In Which Regions is ISO 13485 Applicable?įor most medical devices, compliance to ISO 13485 is required by all European Union members, Canada, Japan, Australia and many other nations. This in turn provides economic benefits in the form of reduced scrap and general process efficiencies. In addition to being a regulatory requirement, an ISO 13485-compliant QMS makes good business sense because it helps device manufacturers minimize variation. The guidelines for maintaining effective quality management processes outlined in ISO 13485 are all geared toward the safe design, manufacture and distribution of effective medical devices. Since different countries often have different standards, ISO 13485 is intended to provide a globally harmonized model of QMS requirements for international markets. It is focused on maintaining QMS effectiveness and meeting regulatory and customer requirements. ISO 13485 is the most common medical device QMS regulatory standard in the world. The aim of this article is to answer frequently asked questions about ISO 13485 manufacturing and the related regulatory requirements that apply to medical device companies’ use of QMS. The ISO 13485 international standard is the world’s most widely used means of measuring the effectiveness of a medical device manufacturer’s quality management system (QMS). But all medical devices have one thing in common: they benefit immensely from being designed and manufactured in alignment with ISO 13485. Others are as simple as a tongue depressor. Some medical devices are as complex as a remote, personalized heart failure sensor.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
